Case of Lung Transplantation in a Recipient with Emphysema: Importance of Determining Alpha 1-Antitrypsin Deficiency Status for Lung Transplantation
Christine M. Bojanowski ![]() , Ian Britton
, Ian Britton ![]() , Kamyar Afshar *
, Kamyar Afshar *![]()
- Division of Pulmonary, Critical Care and Sleep Medicine, University of California, La Jolla, CA 92037, USA
* Correspondence: Kamyar Afshar![]()
Received: April 18, 2018 | Accepted: June 21, 2018 | Published:July 16, 2018
OBM Transplantation 2018, Volume 2, Issue 3 doi:10.21926/obm.transplant.1803015
Academic Editor: Pål Dag Line
Special Issue: Lung Transplant
Recommended citation: Bojanowski CM, Britton L, Afshar K. Case of Lung Transplantation in a Recipient with Emphysema: Importance of Determining Alpha 1-Antitrypsin Deficiency Status for Lung Transplantation. OBM Transplantation 2018;2(3):015; doi:10.21926/obm.transplant.1803015.
© 2018 by the authors. This is an open access article distributed under the conditions of the Creative Commons by Attribution License, which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is correctly cited.
Abstract
Here we present a case of a lung transplant recipient with alpha 1-antitrypsin deficiency as well as conducted a retrospective review of the United Network for Organ Sharing (UNOS) database from 1990 – 2016 specifically grouping patients with alpha-1 antitrypsin deficiency (A1ATD) and chronic obstructive pulmonary disease (COPD).
Keywords
Lung transplant; COPD; emphysema; alpha 1-antitrypsin deficiency
1. Introduction
COPD, when combined with alpha-1 antitrypsin deficiency (A1ATD), accounts for 35% of all transplant recipients [1]. A1ATD remains an under recognized and under diagnosed genetic condition that can predispose individuals to both COPD and liver disease. Despite rigorous recipient candidate testing phase, not all lung transplant centers perform alpha 1-antitrypsin deficiency phenotype and serum value testing to rule out this genetic potential. Lack of appropriate testing subjects these candidates to a great disadvantage both in the pre-transplant and post-transplant phase of their lives. It has been established that patients with A1ATD that have confirmed lung obstruction, have a more rapid decline in lung function compared to alpha 1 sufficient individuals in the pre-transplant phase [2,3,4]. However, the rate of FEV1 decline post lung transplant has not been fully delineated. It is unknown that individuals who are tested and diagnosed with A1ATD who are initiated on alpha 1-replacement should have less acute cellular rejection episodes and a longer post lung transplant survival.
Here we present a case and examine data from the United Network for Organ Sharing (UNOS) and attempt to account for the differences seen in transplant outcomes between A1ATD patients as compared to the COPD population by looking at rates of rejection, airway dehiscence, and survival. The primary reason why the US population was examined is that both testing and alpha -1 replacement therapy is readily available for individuals with severe alpha 1-antitrypsin deficiency.
2. Clinical Vignette
A 69 year-old man with COPD/Emphysema was listed for lung transplantation with a lung allocation score of 35.17. His FEV1 20% predicted with a BODE index of 7. He required 6L/min supplement oxygenation at rest. Fluticasone-salmeterol 500-50 mcg inhaled twice daily and tiotropium 18 mcg inhaled q daily included his inhaler therapies along with azithromycin 250 mg oral daily. He has had 3 exacerbations annually. Alpha 1-antitrypsin phenotype and levels were positive for PiSZ and 56.9 mg/dL respectively. There were no liver abnormalities noted on surveillance testing. He was started on augmentation therapy and several weeks later underwent successful single right lung transplantation. He received induction therapy with basiliximab along with standard triple-drug immunosuppressive medications including tacrolimus, prednisone, and mycophenolate mofetil. The donor was a young male with head trauma and sustained a left lung pneumonia from Serratia and Staph aureus. There were no peri-operative events, presence of primary graft failure or arrhythmias. Exogenous alpha-1 replacement therapy was continued post lung transplantation. By month 3, his FEV1 had reached 72% predicted and by month 12, the FEV1 was maintained at 70% (Figure 1). During the first year, he did not have any episodes of acute cellular rejection. There was only one infectious episode from rhinovirus. Although there was a hyperinflated native left lung, it did not adversely affect him with regards to symptoms or allograft function (Figure 2).

Figure 1 Pulmonary function tests 12 months after undergoing a right single lung transplantation.
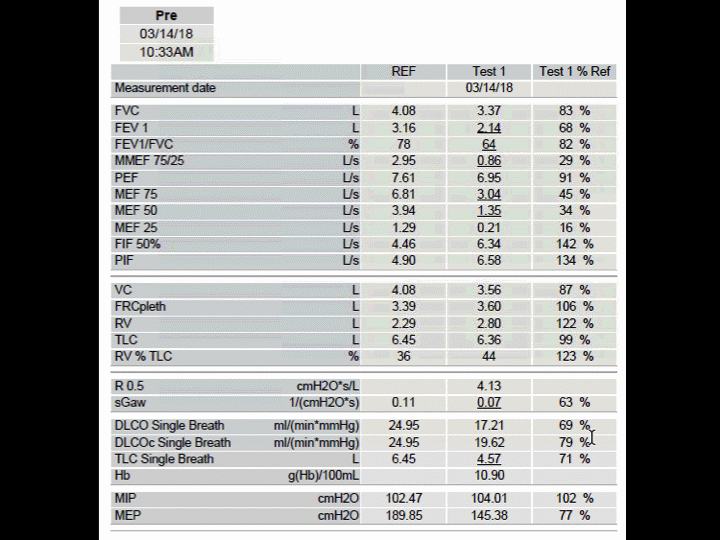
Figure 2 Chest radiograph showing a hyperinflated left native lung COPD and a right single lung allograft.
3. Discussion
It is known that mortality following lung transplantation varies by diagnosis. Poor long-term survival following lung transplantation in individuals with COPD has been attributed to an older aged population. Since not all lung transplant recipients are routinely tested for A1ATD, there is a selection bias in the interpretation of observed death in both groups. A review of the world-wide lung transplants through the Registry of International Society for Heart and Lung Transplantation (ISHLT) shows that the patients with A1ATD tend to do worse immediately after the post-operative period, but that the overall long-term survival surpasses that for patients with COPD. This is similar to the UNOS reports we have reviewed. From January 1, 1990 to February 5, 2016, a total of 28,583 transplants occurred in the United States. 8,769 transplants (30.7%) were performed for patients with COPD while 1,465 transplants (5.13%) were performed for A1ATD. A1ATD lung transplant recipients were younger as compared to COPD lung transplant recipients. (mean age 50.5 years vs. 58 years, p < 0.0001). The gender distribution was rather similar, but there were slightly more men transplanted having A1ATD, while more women were transplanted with COPD as the underlying diagnosis (Table 1). There were a total of 367 recipients with airway dehiscence in both of these groups. Compared to recipient with COPD, A1ATD lung transplant recipients had a higher rate of airway dehiscence (2.32% vs. 0.94%). Short-term patient and graft survival is worst for A1ATD lung transplant recipients, but is more favorable in the long term (Table 2).
Table 1 Number of transplants based on diagnosis at transplant and recipient gender.
|
|
A1ATD |
COPD |
p-value |
|
Female |
574 (4.44%) |
4,511 (34.9%) |
< 0.0001 |
|
Male |
891 (5.69%) |
4,258 (27.2%) |
< 0.0001 |
Table 2 Short-term and long-term patient and graft survival for A1ATD and COPD lung transplant recipients.
|
Patient Survival |
6 months |
1-year |
5-year |
10-year |
|
A1ATD |
85.52% |
79.37% |
53.55% |
30.67% |
|
COPD |
89.41% |
84.32% |
51.61% |
23.92% |
|
|
||||
|
Graft Survival |
6 months |
1-year |
5-year |
10-year |
|
A1ATD |
84.84% |
78.55% |
51.94% |
28.88% |
|
COPD |
88.98% |
83.75% |
50.35% |
23.24% |
In Sweden, a comparison of 83 PiZZ patients with severe emphysema patients who underwent lung transplantation were compared to 70 controlled lung transplant recipients (controlled for age, gender, smoking history and lung function) [5]. The median survival in the A1ATD group was much better at 11 years. One explanation for the worst short- term patient and graft survival is explained by Gulak et al [6]. In this analysis, it was shown that A1ATD lung transplant recipients were more likely to be treated for rejection as compared to COPD (46.6% vs. 42.1%, p=0.04) within the first year of lung transplantation. This group also showed a significantly higher risk of death in the first year [Hazard ratio (HR): 1.46, 95% Confidence interval 1.25-1.71].
In a single center study at the Cleveland Clinic, Banga et al. described the lower lung function findings in 45 A1ATD double lung transplant recipients as compared to 231 COPD counterparts [7]. There was no statistical difference in rejection or survival between the two groups (p=0.09). Despite a younger age of lung transplantation, there was a lower 1year survival (73%) in the A1ATD group when compared to COPD group (86%). An important notation is that only 6 patients with A1ATD had augmentation therapy re-initiated after lung transplant and this was not immediately post transplantation.
Thus far, there has been only 1 reported abstract studying the use of augmentation therapy following human lung transplantation in 5 A1ATD deficient (2 double and 3 single) lung transplant recipients [8]. In the 4 patients included for analysis, stabilization of lung function was noted at 60-90 days after initiation of therapy. The mean time for starting augmentation therapy post lung transplantation was 4.94 years (range 0.68-9.98 years). Prospective studies are still needed to further evaluate the potential use of augmentation post transplant in alpha 1-antitrypsin deficient patients. Recently, the immune modulation function of the alpha 1-antitrypsin protein has been evaluated; particularly in pancreatic (islet) cell transplantation. Alpha 1-antirypsin (A1AT) is a known potent inhibitor of the proteolytic enzymes neutrophil elastase, cathepsin G, proteinase 3 and neutrophil serine proteinase-4 [9]. Animal studies have shown that infusion of recombinant alpha 1-antitrypsin at 90 mg/kg dose prolongs allograft survival in mice through an improved immune tolerance mechanism [10,11]. Iskender and colleagues utilized A1AT to attenuate reperfusion injury after prolonged hypothermic preservation in a large animal lung transplant model [12]. They showed that treatment with A1AT suppressed inflammatory responses in large animals and showed improved graft function post transplant. With respect to earlier administration of exogenous alpha-1 antitrypsin lung transplant population, Gao et al. studied its effects of ischemic-reperfusion injury [13]. Through the use of a pulmonary cell culture model, the effects of A1AT on cytokine production and cell death were examined. They also looked at a rat pulmonary IR model to determine the effects of A1AT on acute lung injury, function, cell death, and inflammatory response. Their results showed that A1AT significantly inhibited cell death and inflammatory cytokine release in a dose-dependent fashion in vitro. In vivo, A1AT administration improved lung oxygenation and reduced pulmonary edema. They also showed that A1AT inhibited neutrophil infiltration into the lung and reduced IR-induced inflammatory mediators in plasma including interleukin (IL)-1α, IL-4, IL-12p70, monocyte chemotactic protein 1, and tumor necrosis factor-α.
4. Conclusions
Routine testing for A1ATD phenotypes and serum levels in patients with COPD should be performed in all lung transplant candidates to better determine the true incidence in the transplant population. Additionally, confirmatory testing will help optimize therapy prior to transplant listing given the longer waitlist times for candidates with obstructive lung disease. Unfortunately certain misconceptions to testing and treating A1ATD create barriers to this. Misconceptions include: 1) recurrence of the emphysematous disease is rarely seen following lung transplantation 2) limited benefit to starting replacement therapy and 3) older individuals with COPD do not have A1ATD as it is a condition attributed to younger patients. Given the potential immunological benefit and reduction of airway inflammation in the continued genetically susceptible population, alpha 1-antitrypsin replacement appears to have preliminary benefits. Prospective randomized control trials should be performed to assess the short term and long term benefits of alpha-1 antitrypsin replacement therapy following lung transplantation.
Author Contributions
All authors contributed to the background information and writing/editing of the manuscript.
Competing Interests
The authors have declared that no competing interests exist.
References
- Yusen RD, Edwards LB, Dipchand AI, Goldfarb SB, Kucheryavaya AY, Levvey BJ, et al. The registry of the international society for heart and lung transplantation: thirty-third adult lung and heart–lung transplant report—2016; Focus theme: primary diagnostic indications for transplant. J Heart Lung Transpl. 2016; 35: 1170-1184. [CrossRef]
- Stockley RA, Edgar RG, Pillai A, Turner AM. Individualized lung function trends in alpha-1-antitrypsin deficiency: a need for patience in order to provide patient centered management? Int J Chronic Obstr. 2016; 11: 1745. [CrossRef]
- Pott GB, Chan ED, Dinarello CA, Shapiro L. α‐1‐Antitrypsin is an endogenous inhibitor of proinflammatory cytokine production in whole blood. J Leukocyte Biol. 2009; 85: 886-895. [CrossRef]
- Jonigk D, Al-Omari M, Maegel L, Müller M, Izykowski N, Hong J, et al. Anti-inflammatory and immunomodulatory properties of α1-antitrypsin without inhibition of elastase. P Natl Acad Sci. 2013; 110: 15007-15012. [CrossRef]
- Tanash HA, Riise GC, Hansson L, Nilsson PM, Piitulainen E. Survival benefit of lung transplantation in individuals with severe α1-anti-trypsin deficiency (PiZZ) and emphysema. J Heart Lung Transpl. 2011; 30: 1342-1347. [CrossRef]
- Gulack B, Ganapathi A, Speicher P, Chery G, Snyder L, Davis R, et al. Survival after lung transplant in alpha-1-antitrypsin deficiency recipients compared to other forms of chronic obstructive pulmonary disease. J Heart Lung Transpl. 2015; 34: S243-S244. [CrossRef]
- Banga A, Gildea T, Rajeswaran J, Rokadia H, Blackstone EH, Stoller JK. The natural history of lung function after lung transplantation for α1-antitrypsin deficiency. Am J Resp Crit Care. 2014; 190: 274-281.
- Ravichandran B, Feist A, Magana M, Reed S, Lane J, Yung G. Alpha-1 Proteinase Inhibitors post-lung transplantation: a single center experience. J Heart Lung Transpl. 2013; 32: S295. [CrossRef]
- Stockley RA. The multiple facets of alpha-1-antitrypsin. Ann Transl Med. 2015; 3.
- Lewis EC, Mizrahi M, Toledano M, DeFelice N, Wright JL, Churg A, et al. α1-antitrypsin monotherapy induces immune tolerance during islet allograft transplantation in mice. P Natl Acad Sci. 2008; 105: 16236-16241. [CrossRef]
- Lewis EC, Shapiro L, Bowers OJ, Dinarello CA. α1-Antitrypsin monotherapy prolongs islet allograft survival in mice. P Natl Acad Sci USA. 2005; 102: 12153-12158. [CrossRef]
- Iskender I, Sakamoto J, Nakajima D, Lin H, Chen M, Kim H, et al. Human α1-antitrypsin improves early post-transplant lung function: Pre-clinical studies in a pig lung transplant model. J Heart Lung Transpl. 2016; 35: 913-921. [CrossRef]
- Gao W, Zhao J, Kim H, Xu S, Chen M, Bai X, et al. α1-Antitrypsin inhibits ischemia reperfusion-induced lung injury by reducing inflammatory response and cell death. J Heart Lung Transpl. 2014; 33: 309-315. [CrossRef]



