Application of Microfluidic Biochips for Human Islet Transplantation
Julianna Holzer 1 ![]() , John Wu 2
, John Wu 2 ![]() , Yi He 1
, Yi He 1 ![]() , Mingyang Ma 1
, Mingyang Ma 1 ![]() , Yuan Xing 1
, Yuan Xing 1 ![]() , Jose Oberholzer 1
, Jose Oberholzer 1 ![]() , Yong Wang 1, *
, Yong Wang 1, * ![]()
- Department of Surgery/Transplant, University of Virginia, Charlottesville, Virginia, USA
- Albemarle High School, 2775 Hydraulic Rd, Charlottesville, Virginia, 22901, USA
Academic Editor: Yasuhiko Sugawara
Special Issue: Current Advancement of Islet Cell Transplantation in the Treatment of Diabetes Mellitus
Received: November 08, 2018 | Accepted: December 18, 2018 | Published: December 21, 2018
OBM Transplantation 2018, Volume 2, Issue 4 doi:10.21926/obm.transplant.1804034
Recommended citation: Holzer J, Wu J, He Y, Ma M, Xing Y, Oberholzer J, Wang Y. Application of Microfluidic Biochips for Human Islet Transplantation. OBM Transplantation 2018; 2(4): 034; doi:10.21926/obm.transplant.1804034.
© 2018 by the authors. This is an open access article distributed under the conditions of the Creative Commons by Attribution License, which permits unrestricted use, distribution, and reproduction in any medium or format, provided the original work is correctly cited.
Abstract
In this review, we discuss the application of microfluidic devices in studying the physiology and pathophysiology of human islets and beta-cells, especially its application for human islet transplantation. Human islet transplantation is a promising therapy for Type I diabetes; however, the islet transplant outcomes for achieving complete insulin independence are far from perfect and face many challenges. This review focuses on the microfluidic devices developed in our laboratory, which can address these challenges in the field of human islet transplantation, and briefly review the design and fabrication principles of these microfluidic devices and their main application.
Keywords
Microfluidics; pancreatic islet of Langerhans; islet perifusion; islet physiology; human islet transplantation
1. Introduction
Type I diabetes mellitus (T1DM) is a devastating disease caused by the auto-immuno destruction of β-cells in the islets of Langerhans. T1DM currently affects 1.25 million Americans, including 200,000 youth. Five million people are expected to have T1DM by 2050, including nearly 600,000 youth. The T1DM-associated annual healthcare cost in the US is about 14 billion.
Since the Edmonton Protocol was introduced in 2000 [1], islet allo-transplantation has become a promising therapy for T1DM, especially for those with frequent hypoglycemia unawareness, who make up 10% of the T1DM population. The transplanted islets can physiologically regulate insulin and glucagon secretion in response to instantaneous blood glucose changes, which provides superior glycemic control compared to insulin injection in a patient with frequent hypoglycemia unawareness. Islet transplantation has many advantages over whole pancreas transplantation for the treatment of T1DM, since it involves only a minor surgical procedure with much lower morbidity and mortality rates [1,2]. However, the therapy has been shown to have varying results for short and long-term insulin independence [3,4], which is related to inconsistent transplant islet quality, peri-transplant islet graft failure associated with instant blood mediated inflammatory reaction (IBMIR), immunotoxicity, an unideal transplant site (hypoxia and high immunosuppressant concentration in the portal system), and islet exhaustion. Although 60-100% of patients can achieve insulin independence in the majority of transplant programs, the rate can drop to as low as 10% between 2 and 5 years, even when a significant number of islets (>10000 IEq/kg) are transplanted from 2 or more donors [5].
2. Microfluidic Technology
Microfluidic technology is a special class of Bio-Microelectromechanical systems (Bio-MEMS). This technology has been emerging as a valuable tool for a wide range of biological applications. The general advantages of microfluidic tools in pancreatic islet research have been reviewed previously by our group [6].
In brief, microfluidic technology has the following advantages: reagent and analyst consumptions are minimal, the allowance for leveraging of microscale flow phenomena such as laminar flow and rapid diffusion on the small scale, and easier implementation of new experimental modalities currently not possible with macroscale tools. In addition, multiple tasks can be integrated onto a single device to improve experimental throughput. More than 10,000 microfluidics papers have been published over the last 20 years. To date, the application of microfluidics for islet and/or beta-cell studies is very limited, as only a handful of laboratories in the United States have pursued this research [7,8,9,10,11,12,13,14].
2.1 The Principle of Microfluidic Device Design and Fabrication
Detailed reviews on microfluidic design and fabrication for islet have been described elsewhere [6,15]. Microfluidic techniques entail the design, fabrication, and application of a specific device for the manipulation of fluids at the microscale level. Typically, ‘micro’ entails one of the following features: small volume (often in the scale of nl, pl, fl), small size, low energy consumption, or the use of some physical aspect of the microdomains, such as size, laminar flow, or rapid diffusion.
Several different materials have been used for microfluidic fabrication including silicon, metal, glass, and various polymers. Since all studies discussed in this review use polydimethylsiloxane (PDMS) for device fabrication, only the fabrication of PDMS devices is briefly described.
In general, PDMS microfluidic devices are designed using computer-aided design (CAD) software such as AutoCAD and then printed at a high-resolution (16,000 dpi) on a transparent film. The film is used as a photomask to selectively crosslink photoresist (SU-8) pre-spun to a desired thickness on a silicon wafer. Once coated, the wafers undergo pre-exposure baking at 65°C for 5 minutes, 90°C for 2 hours, and then 65°C for 1 minute. Finally, the high-resolution photomask is placed in contact with the photoresist-covered wafer and is irradiated with 365 nm filtered UV radiation to initiate crosslinking. The irradiated wafer is further cross-linked on a hotplate at 95°C for 1 hour and then placed in SU-8 developer solution, where unpolymerized SU-8 is dissolved. The completed master is then cleaned with isopropyl alcohol and water, dried with compressed nitrogen gas, and dehydration baked. After completing the master, PDMS is prepared for molding/soft-lithography. PDMS precursor (usually Sylgard 184) and cross-linking agent are added to a weighing boat at a 10:1 ratio by mass and thoroughly mixed. A vacuum chamber is next used to extract any bubbles, and the PDMS mixture is then poured over the SU8 master. After overnight curing at room temperature or 2 hours on an 85ºC hotplate, the PDMS is completely cured, retaining the channel structures from the master. A complete microfluidic device is comprised of multiple layers, each requiring a unique master mold. After the PDMS is cured, each layer is cut from the bulk PDMS and inlet and outlet ports are punched into the appropriate layer. The PDMS layers are then bonded under plasma treatment into a working device.
2.2 Microfluidic Device for Evaluation of Human Islet Function and Viability
As aforementioned, human islet transplant outcomes vary, which is mainly due to the inconsistent quality of human islet preparations transplanted. Despite standardization and the application of good manufacturing principles in the islet isolation process, lot-to-lot variability cannot be avoided. To reduce the risk of transplanting low quality islets, appropriate product release testing is needed. While tests for sterility, identity, and purity are well established, no reliable method for the assessment of islet potency exists.
An ideal potency test should predict islet graft function after transplantation. Presently, potency tests rely mostly on the assessment of cellular viability. In general, viability tests evaluate the metabolic activity or the integrity of the cell or nuclear membrane, and are not specific to a particular cell type within islets. Glucose-stimulated insulin secretion is the most relevant function of islets and is β-cell specific. Currently, insulin secretion is measured by simple static glucose incubation, which exposes a sample of islets to low and high glucose and determines the basal and stimulated insulin concentrations in the supernatant. However, results of static incubation do not correlate with clinical outcomes such as islet graft function and insulin independence [16].
In vivo potency assays, evaluated by transplanting human islets into immunodeficient nude mice, correlate with clinical transplant outcomes and are currently the gold standard for evaluating islet potency [17] . However, this in vivo analysis takes several weeks to complete; the retrospective indication of islet function renders this assay impractical as a pre-transplant assessment in a clinical setting.
A variety of other tests have been developed to assess islet potency and viability, including measurement of the oxygen consumption rate (OCR) [18,19], reactive oxygen species [20,21], ADP/ATP ratios [22], as well as mitochondrial integrity evaluation [20]. In addition to being only retrospectively useful and lacking predictive value of islet graft function, these assays have multiple limitations. For instance, OCR and ROS assays are mostly conducted statically and have low β-cell specificity, as β-cells only make up 65-80 % of a human islet cell population. Additionally, ROS species are highly reactive with a short physiological half-life of 0.1-5 seconds. Finally, ADP/ATP assays require islet dissociation. Accumulating evidence indicates that chemical and/or ion intra-islet communication between β-cells is important for insulin secretion regulation. Furthermore, the gap junction complexes between adjacent islet cells facilitate inter-islet cell-to-cell communication and help coordinate hormonal output.
Allogeneic pancreatic islets are defined as a biological drug by the FDA. As such, islets cannot be used clinically without an Investigational New Drug application or an approved biologics license application. For islet transplantation to become an FDA licensed product, a well-established islet preparation process and product manufacturing consistency will need to be demonstrated.
Islets of Langerhans are made up of at least five different cell types. Approximately 65–80% of these cells are beta-cells that secrete insulin for maintaining glucose homeostasis. Insulin release displays biphasic and pulsatile profiles in response to glucose. As seen in Figure 1, glucose enters beta-cells via GLUT2, and glycolysis generates pyruvate, which enters the TCA cycle. Pyruvate produces ATP and subsequently closes ATP-sensitive Potassium (KATP) channels, initiating plasma membrane depolarization and causing an increase in intracellular calcium concentration ([Ca2+]i) via voltage-dependent calcium channels (VDCCs). Finally, the glucose-induced [Ca2+]i triggers the fusion of insulin granules to the plasma membrane, resulting in the exocytosis of insulin. The first phase of the biphasic profile corresponds to a prompt, marked insulin secretion (4-8 minutes). With continuous glucose stimulation, a secondary phase consisting of a gradual insulin increase occurs. Alternate pathways of glucose-stimulated insulin secretion, independent of either KATP channels or [Ca2+]i, have been described, but play an insignificant role in insulin secretion.
In 2009, we first designed a three-layer PDMS device integrated with fluorescence imaging of key insulin stimulator secretion coupling factors and insulin secretion kinetics for evaluation of human islet function and viability [23]. The top layer of the device has an inlet and an outlet channel (500 μm in height and 2 mm in width). The middle layer is a perifusion chamber (7 μm in diameter and 3 μm in depth). The bottom layer (150 μm in height) contains microwells for islet immobilization, which are 500 μm in diameter and 100 μm apart (Figure 2A). The device is capable of holding multiple islets while allowing the islets to passively sit without the need for islet fixation. As shown in Figure 2B, a CFD-GEOM computer simulation demonstrates uniform flow distribution in the perifusion chamber with most of the flow reaching the bottom, where islets are trapped in microwells without significant fluid shunting[24]. In the perifusion chamber, various chemical gradients can be created and maintained with a high level of complexity and consistency (Figure 2C) [24]. In addition to serving as an islet perifusion apparatus, fluorescence-based analytical approaches are integrated, significantly increasing its analytical power with good spatiotemporal resolution of the measured parameters (Figure 2D).
We have applied the microfluidic biochip to evaluate 150 human islet preparations and showed that the microfluidic parameters (calcium influx, mitochondrial potentials, and insulin secretion kinetics) can provide better predictive values for in vivo islet graft function and viability in a nude mouse transplant model (data now shown). Further clinical evaluation for predicting human islet in vivo graft function is under investigation.
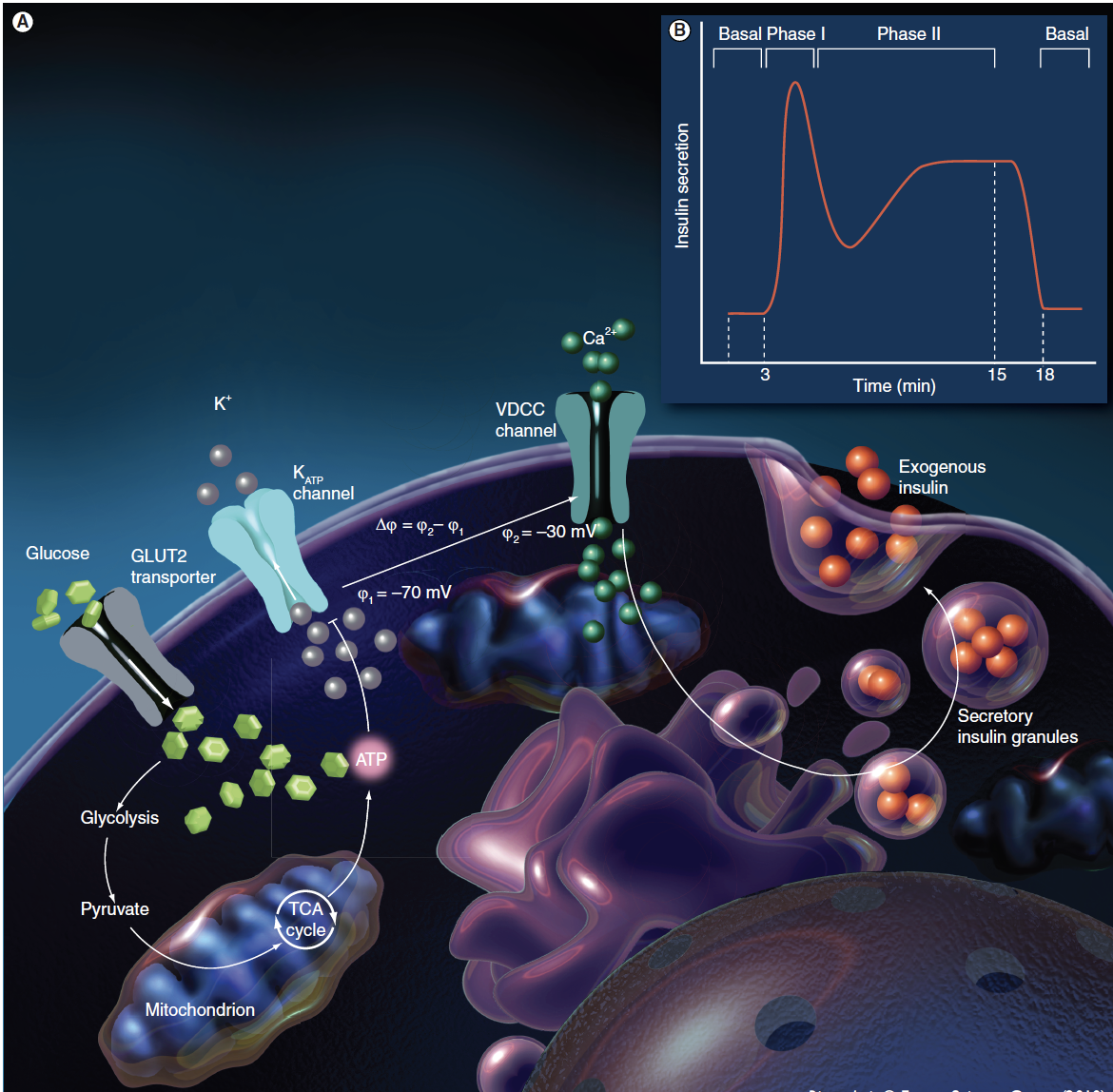
Figure 1 Glucose-induced beta-cell insulin secretion. (A): KATP-dependent insulin secretion pathway. (B): Insulin biphasic secretion kinetics. Adapted with permission from [6].
Microfluidic Islet Array for High Content and High Resolution Imaging
A High-content screening (HCS) platform integrated with analytical tools has become a useful tool in biological research and therapeutic discovery. However, its application for islet study is very limited.
Two of the major challenges of our previous device are the limited number of islets (~50 islets) that can be assessed and the inability to assess the heterogeneous properties of individual islets. Examination of islet heterogeneous properties often provides more detailed physiological information than averaging-based methodologies. It enables a better understanding of human islet functionality that provides a better predictive value for islet transplant outcome. For this purpose, we designed novel microfluidic islet arrays, based on the hydrodynamic trapping mechanism for islets and microencapsulated islets.

Figure 2 First generation of microfluidic Islets-On-Chip. (A): Microfluidic device design principle. (B) Computer simulation of flow velocity and chemical diffusion. (C) Creation of chemical gradients in perifusion chamber. (D) Multiparametric assays for intracellular calcium, mitochondrial potentials, and insulin secretion. Adapted with permission from [23,24].
The microfluidic islet array is a one-layer PDMS device as shown in Figure 3A [25]. The array device utilizes the hydrodynamic trapping principle to immobilize islets. The device contains an array consisting of 2 rows and 10 columns. In each column, there are a total of 300 traps. The trapping site design is a U-cup shaped pocket (250 μm in diameter and 275 μm in depth), superimposed onto a loop channel that is used for delivery of fluids and islets. There is a cross-flow channel (45 μm in width) at the apex of the U-shape pocket. Due to the flow resistance difference between the U-shape cup and the loop channel, the flow encounters less resistance in the unoccupied U-shape cup and when solution takes an islet to the gap between the U-shape cup and the main channel, an islet becomes trapped in the U-shape cup. The trapped islet results in increased resistance in the U-cup, which redirects the flow into the loop channel. The COMSOL fluid-flow simulation results were depicted in Figure 3B, showing that the Q1 velocity is much larger than the Q2 velocity, leading islet to be trapped in the U-cup pocket.
We further characterized the impact of geometries on trapping efficacy. Our results showed in Figure 3B that a Q1/Q2 ratio of 5.5 (a high resistance ratio) led to multiple islets per site. When the Q1/Q2 was equal to 0.7, the solution going into the gap was not enough for optimal loading. By modifying the resistance of the straight channel (Q1/Q2 = 2.8), single islet occupancy per trap was achieved. At this ratio of 2.8, 99% ± 2.5% of the sites were filled, with 95% ± 2% of the filled sites having an individual islet.
Together with real-time fluorescence imaging, the array can be used for islet flow cytometry to track the dynamic physiological and pathophysiological behavior of individual islets. Heterogeneous responses of intracellular calcium and mitochondrial potential changes from isolated human islets are shown in Figures 3C. Human islets displayed heterogeneous calcium profiles in response to glucose and KCI (25 mM glucose: 144.2% ± 1.74, Max: 149.9%, Min: 139.9%; 30 mM KCI: 176.2% ± 1.54, Max: 179.1%, Min: 173.5%), and heterogeneous mitochondrial potential changes in response to glucose (25 mM glucose: 71.3% ± 2.02, Max: 76.8%, Min: 62.3%). The heat-map of calcium concentration and mitochondrial potential changes is depicted in Figure 3C.

Figure 3 Microfluidic islet array for high content and high resolution assay. (A): Microfluidic islet array and design. (B) Islet loading optimization. (C) Fluorescence imaging of calcium influx and mitochondrial potentials change in response to glucose or KCI and heat-maps of calcium influx and mitochondrial potentials changes. Adapted with permission from [25].
Islet transplantation provides tight glycemic control for T1DM, but requires immunosuppression that has unwanted side effects on both the patient and islet graft. The immunoisolation of islets in microcapsules has been heavily investigated to avoid immunosuppression. If successful, this strategy would have a significant impact on islet transplant outcomes. Despite initial promising results in both small animal and nonhuman primate transplant models, only short-term and partial graft function has been achieved in clinical trials. Factors proposed to be responsible for graft loss include insufficient material biocompatibility, limited immunoprotective properties, hypoxia, and suboptimal insulin release. While research has focused heavily on material biocompatibility and immune-protection, a comprehensive understanding of physiological changes of microencapsulated islets is often constrained due to the availability of research tools.
For this reason, we developed a three-layer microfluidic array for microencapsulated islets based on the same hydrodynamic trapping principle (Figure 4A and B) [26]. In addition, an oxygenation channel was integrated to study the pathophysiological changes under hypoxia, one factor contributing to the graft failure of microencapsulated islet. Oxygenation efficacy in the microfluidic channel was evaluated in both gaseous (diffused) and aqueous (dissolved) modes (Figure 4C). In the diffusive mode, the device was capable of creating and maintaining the targeted oxygen concentrations with high consistency. In cyclic oxygenation protocol (21-5-21%), the time needed to switch from 21% to 5% oxygen concentration was less than 40 seconds as was the reversion back to 21% from 5%. Equally important, both concentrations were able to be well-maintained overtime (21.21% ± 0.05 and 6.22% ± 0.03). Similarly, the time needed to change from one oxygen concentration to another in step-down protocol was also less than 40 seconds, and again, was well-maintained overtime (21.33% ± 0.04, 11.53% ± 0.05, 6.33% ± 0.02, and 1.77% ± 0.02, respectively). In the dissolved mode, the time needed to switch from 21% (22.07% ± 0.13) to 5% (6.83% ± 0.08) was approximately 120 seconds, which was longer than the diffusion mode. Similarly, the time needed to change from a particular oxygen concentration to another in the step-down protocol was less than 120 seconds and was stably maintained (20.87% ± 0.06, 11.63% ± 0.03, 6.45% ± 0.03, and 1.68% ± 0.03, respectively).

Figure 4 Microfluidic encapsulated islet array for high content and high resolution assay. (A) Design principle and dimension for the three-layer array. (B) Hydrodynamic trapping computer simulation. (C) Oxygen modulation. Adapted with permission from [26].
As previously explained, hypoxia is widely considered to be one of the primary factors associated with the functional loss of encapsulated islets and has also been linked to the immunoisolation failure of microencapsulation devices. Isolated islets are exposed to a hypoxic environment at the following levels: (i) isolated islets have a disrupted vascular network and are dependent on diffusion for their oxygen supply, the microencapsulation process further aggravates islet hypoxia by both preventing islet revascularization that normally occurs and increasing oxygen diffusion distance due to capsule sizes that are often larger than 500 μm; (ii) the intraperitoneal space, a common transplant site for microencapsulated islets, has a low oxygen tension, where the O2 concentration is approximately 3.5-5%, which is significantly lower than the in situ pancreas. In addition to function loss, hypoxia may also facilitate the attraction of macrophages and subsequently cause cell overgrowth on the microcapsule surface.
As shown in Figure 5A, the calcium influx changes of microencapsulated human islets in response to 25 mM glucose challenges were oxygen concentration-dependent and were inhibited by hypoxia [26]. Under normoxia, the average intracellular calcium concentration increased by 10% ± 4.16 in response to 25 mM glucose stimulation, while hypoxic concentrations decreased intracellular calcium responses: 8.19% ± 2.5 in 10% O2, 3.57% ± 1.18% in 5% O2, and 1.70% ± 0.64 in 1% O2 (p < 0.01 when 21% vs. 5% and 1%, as well as p < 0.01 when 10% vs. 5% and 1%). Similarly, mitochondrial potential changes, often used as an indicator of cellular energetic status, were also inhibited in an oxygen concentration-dependent manner (21-10-5-1%: 17.23% ± 3.13, 8.83% ± 3.53, 6.40% ± 2.56, and 4.09% ± 1.37 (p < 0.01 when comparing 21% vs. 10, 5, and 1%; p < 0.01 when comparing 10% vs. 5% and 1%) as shown in Figure 5B.
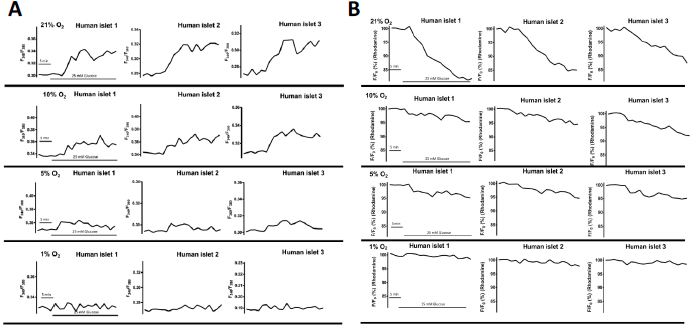
Figure 5 Hypoxia impaired intracellular calcium signaling and mitochondrial potentials of encapsulated human islets in response to glucose. (A) Representative traces of calcium influx of encapsulated human islets in response to 25mM glucose. (B) Representative traces of mitochondrial potential changes of encapsulated human islets in response to 25mM glucose. Adapted with permission from [26].
This device achieved a high trapping efficacy for microencapsulated islets (∼99%) with minimal physical stress on islets (data not shown). The integration of the gas modulation allowed for rapid membrane-diffused oxygenation of islets at the microscale-level. This provided a practical tool to study hypoxia on microencapsulated islets. This is first report on real-time multiparametric imaging of metabolic changes of microencapsulated islets under hypoxia, a feat previously unachievable using either hypoxic chambers or existing microfluidic devices.
Development of microfluidic-based arrays as suitable high-content screening (HCS) platforms will be beneficial not only for studying the physiology of islets and microencapsulated islets, but also for beta-cell-like stem-cell characterization and therapeutic development.
2.3 Integration of Pumpless Flow Delivery in Microfluidics
Syringe pumps, often used in microfluidics for fluid delivery, are bulky, expensive, and inconsistent with the simplistic philosophy of microfluidics. Accessory components (syringe and tubing) can increase reagent consumption, create bubbles, and reduce spatiotemporal parameters resolution. Significant efforts have been made to integrate microscale pumps within a microfluidic chip. However, complex design and fabrication processes, as well as the need for an external power source, averts their applications [27].
The concept of a passive-driven pump has also been proposed [28]. Ideally, a passive-driven pump should be simple and easy in design, fabrication, integration, and operation. Surface-tension-based pumpless systems in microfluidics are one type of passive-driven pump in which flow is driven by surface tension differences due to the different diameters of the inlet and outlet [29]. According to the Young-Laplace equation, a smaller curvature between the fluid droplet and the microfluidic surface will cause a larger pressure, which leads the fluid to flow from the smaller diameter inlet toward the larger diameter outlet. The curvature of the droplet is determined by the hydrophobicity of the surface. The equation, expressed as ![]()
![]() , is a nonlinear partial differential equation that describes the capillary pressure difference between two static fluids in which the gravitational and viscous forces are negligible because the Bo number (Bo = ρgR2/γ ≈ 0.01) and capillary number (Ca = μv/γ ≈ 10-4) are extremely small. Therefore, surface tension dominates in driving fluid flow. When Ro >> Ri, the backward pressure generated from the outlet can also be neglected, which prevents back flow and turbulence
, is a nonlinear partial differential equation that describes the capillary pressure difference between two static fluids in which the gravitational and viscous forces are negligible because the Bo number (Bo = ρgR2/γ ≈ 0.01) and capillary number (Ca = μv/γ ≈ 10-4) are extremely small. Therefore, surface tension dominates in driving fluid flow. When Ro >> Ri, the backward pressure generated from the outlet can also be neglected, which prevents back flow and turbulence
We have designed a pumpless prototype based on surface tension principles and further integrated an islet array, which holds individual islets [27]. The device has an inlet with a Ri of 1.5 mm and an outlet with a Ro of 7.5 mm (Ro / Ri = 5) (Figure 6A). The islet array is based on the hydrodynamic trapping principle, as described earlier. The total channel length is about 60 mm with a total volume of about 10 µL.
The computational simulation of flow velocity using COMSOL multiphysics showed a bell-shaped flow velocity profile with the highest velocity in the channel center (23.5 mm/s). The flow velocity was reduced gradually near the channel wall due to increased resistance. A comparison of the simulation and experimental results demonstrated a close similarity between the distributions of velocity (Figure 6B). Since the distance between the inlet and the trap area was only about 8 mm, it took approximately 0.3 seconds to get the cells stimulated and took approximately 2.5 seconds for a complete flow exchange. The rate of solution exchange was significantly faster as a result of small channel size and volume compared to our previous perifusion chamber design, which required 2-3 minutes to fully exchange solution. Overall, the fast flow delivery would provide an efficient stimulation for islet cells.

Figure 6 The surface tension driven pumpless microfluidic for islets. (A) Working principle of the surface tension driven flow delivery, schematic of the pumpless device, and device dimension. (B) Computer simulation of flow rat in the microchannel. Adapted with permission from [27].
Adjustable flow speeds can be achieved by designing microfluidics with different Ri/Ro ratios. When the inlet and outlet are the same size, no fluid flow was observed due to zero pressure difference between the inlet and outlet. The average flow rates increase if the dimension difference between Ri and Ro is increased (data not shown).
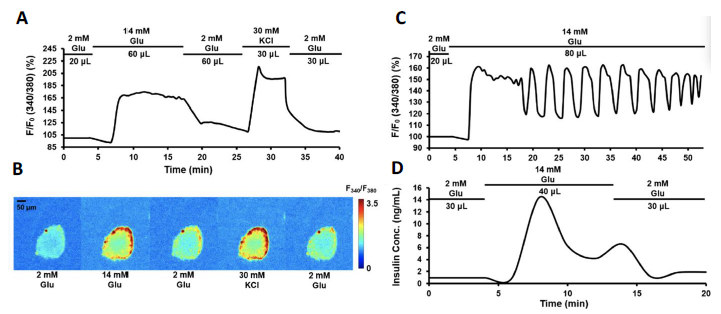
Figure 7 Perifusion and imaging of rodent islets. (A) Representative trace of rodent islet calcium signaling in response to 14 mM glucose and 30 mM KCl. (B) Images of Fura 2340/380 in corresponding to each stimulation phase. (C) Representative trace of rodent islet calcium oscillation in response to 14 mM glucose. (D) Representative trace of rodent islet insulin secretion in response to 14 mM glucose. Adapted with permission from [27].
As shown in Figure 7, integration of the islet array and pumpless delivery system significantly reduced total flow volume (37.5 mL to 0.18 mL). Stimulation protocol is also reduced from 70 minutes to 25 minutes. With collection of perifusate at 20 μL/min (instead of 250 μL/min), the spatiotemporal resolution of calcium biphasic signaling was well preserved and improved, as well as the insulin secretion kinetics. Additionally, it is much easier to operate and is portable.
3. Chanllenges and Future Direction
In the last couple of decades, microfluidics has crisscrossed almost every corner of biological sciences, with strong promise of more efficient device performance and potential clinical application. Initial publications articulating novel technological innovations are beginning to be supplanted to some extent by new biological or chemical science. Microfluidic systems have come a long way toward fulfilling many of the early predictions regarding performance, utility, and functionality. However, there are many challenges that need to be addressed before this technology becomes developed enough to be accepted by researchers in commonplace laboratory applications, such as biocompatibility of microfluidic-building materials, stability of biological entities in the constrained microenvironments, and inherent challenges of handling complex molecules in narrow confinements. Furthermore, fundamental understanding on fluid mechanics and transport phenomena over small scales as well as technological advancements on fabrication, process control, and optical diagnostics also play a key role towards achieving that feat.
The microfluidic application in the field of diabetes research and treatment has undergone significant developments since its formal inception in the early 2000s. Similarly, there are also many challenges of microfluidic application in the area. For example, beta-cell glucose sensing and insulin secretion are very sensitive to flow dynamics and shear forces. PDMS can undergo a degree of shrinkage during curing, and this shrinkage can induce alignment registration problem and then greatly impact flow dynamics, which is very important for multilayer devices. Designing systems with large alignment tolerances may limit the potential for miniaturization, and not be desirable in many applications. One common approach to solve this problem is to characterize the degree of PDMS shrinkage PDMS and scale the master size accordingly. This approach has been successfully used to create functional large-area microfluidics. However, such approach only be feasible for the devices that can tolerate large misalignments. Furthermore, the degree of shrinkage of PDMS is not constant but dependent on cure temperature and time, PDMS component ratios, and layer thickness. These parameters introduce further issues with the characterize-and-scale approach to the shrinkage-induced alignment registration problem. Curing temperatures and environmental parameters must be strictly controlled in order to achieve a consistent degree of shrinkage. Since the amount of shrinkage is dependent on layer thickness, a single calibration result cannot be used in the fabrication of multiple prototype designs. Thus, individual calibration experiments need to be performed for each design. Furthermore, shrinkage calibration techniques will never provide a perfectly accurate scaling factor, and while small errors may be acceptable for low-throughput, proof-of-concept devices, this technique cannot be scaled up to larger-area devices with high feature densities, without extensive time-consuming and iterative calibration studies.
Many in vitro human islet functional assays pre transplant have been developed in the past decades; however, no such assay has been used to precisely predict in vivo islet survival and function, especially for clinical transplant outcomes. The transplanted islet graft function is dependent on in vivo islet cell survival, transplant site environment, beta-cell immunotoxicity, and the revascularization process that may take 2-3 weeks to initiate and months to complete. In order to apply for clinical application, extreme caution needs to be taken for any in vitro functional assay and need to be further verified in both animal transplant model and clinical trials.
This mini review highlights how microfluidic technology is currently used in the field of diabetes research and treatment. Naturally, such a limited review is biased by personal experience and expectation, and as a result, we are likely to have omitted many important applications and uses of microfluidic tools developed by others. Regardless, a primary intention in writing this article was to provide some highlights on the microfluidics developed in our laboratory. Although microfluidics can provide the experimentalist with many advantages, it is not a cure-all. Indeed, many features of microfluidic systems that were once deemed critical to their use or adoption are now less compelling to attendant improvements and refinements in conventional technologies. A microfluidic tool must make a persuasive case for adoption on the basis of factors such as analytical performance, usability, and information yield. For example, development of online integrated hormone assay will facilitate into more robust and efficient multiparametric assay for islet cells. A rather obvious but important challenge to the diabetes community is to recognize that microfluidic tools are not unequivocally enabling in many situations and that the ability to identify the right applications, which find immediate benefit when leveraging microfluidic control, will be critical in fostering more widespread adoption of microfluidic technology.
Author Contributions
Each of the authors contributed partially in the manuscript writing. JO and YW are responsible for final approval.
Funding
This study was supported by the Chicago Diabetes Project (CDP) and the National Institute of Health (NIH) R01 DK091526 and R25 DK105924-01.
Competing Interests
The authors have declared that no competing interests exist.
References
- Shapiro AMJ, Lakey JRT, Ryan EA, Korbutt GS, Toth E, Warnock GL, et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med. 2000; 343: 230-238. [CrossRef]
- Gangemi A, Salehi P, Hatipoglu B, Martellotto J, Barbaro B, Kuechle JB, et al. Islet transplantation for brittle type 1 diabetes: the UIC protocol. Am J Transplant. 2008; 8: 1250-1261. [CrossRef]
- Qi M, Kinzer K, Danielson KK, Martellotto J, Barbaro B, Wang Y, et al. Five-year follow-up of patients with type 1 diabetes transplanted with allogeneic islets: the UIC experience. Acta Diabetol. 2014; 51: 833-843. [CrossRef]
- Barton FB, Rickels MR, Alejandro R, Hering BJ, Wease S, Naziruddin B, et al. Improvement in outcomes of clinical islet transplantation: 1999-2010. Diabetes Care. 2012; 35: 1436-1445. [CrossRef]
- Hering BJ, Clarke WR, Bridges ND, Eggerman TL, Alejandro R, Bellin MD, et al. Phase 3 Trial of Transplantation of Human Islets in Type 1 Diabetes Complicated by Severe Hypoglycemia. Diabetes Care. 2016; 39: 1230-1240. [CrossRef]
- Wang Y, Lo JF, Mendoza-Elias JE, Adewola AF, Harvat TA, Kinzer KP, et al. Application of microfluidic technology to pancreatic islet research: first decade of endeavor. Bioanalysis, 2010. 2(10): p. 1729-1744. [CrossRef]
- Roper MG, Shackman JG, Dahlgren GM, Kennedy RT. Microfluidic chip for continuous monitoring of hormone secretion from live cells using an electrophoresis-based immunoassay. Anal Chem. 2003; 75: 4711-4717. [CrossRef]
- Shackman JG, Dahlgren GM, Peters JL, Kennedy RT. Perfusion and chemical monitoring of living cells on a microfluidic chip. Lab Chip. 2005; 5: 56-63. [CrossRef]
- Dishinger JF, Kennedy RT. Serial immunoassays in parallel on a microfluidic chip for monitoring hormone secretion from living cells. Anal Chem. 2007; 79: 947-954. [CrossRef]
- Zhang X, Roper MG. Microfluidic perfusion system for automated delivery of temporal gradients to islets of Langerhans. Anal Chem. 2009; 81: 1162-1168. [CrossRef]
- Zhang X, Grimley A, Bertram R, Roper MG. Microfluidic system for generation of sinusoidal glucose waveforms for entrainment of islets of Langerhans. Anal Chem. 2010; 82: 6704-6711. [CrossRef]
- Rocheleau JV, Walker MG, Head WS, McGuinness OP, Piston DW. Microfluidic glucose stimulation reveals limited coordination of intracellular Ca2+ activity oscillations in pancreatic islets. Proc Natl Acad Sci U S A. 2004; 101: 12899-12903. [CrossRef]
- Easley CJ, Benninger RK, Shaver JH, Head WS, Piston DW. Rapid and inexpensive fabrication of polymeric microfluidic devices via toner transfer masking. Lab Chip. 2009; 9: 1119-1127. [CrossRef]
- Varhue WB, Langman L, Kelly-Goss M, Lataillade M, Brayman KL, Peirce-Cottler S, et al. Deformability-based microfluidic separation of pancreatic islets from exocrine acinar tissue for transplant applications. Lab Chip. 2017; 17: 3682-3691. [CrossRef]
- Heileman KL, Daoud J, Tabrizian M. Elaboration of a finite element model of pancreatic islet dielectric response to gap junction expression and insulin release. Colloids Surf B Biointerfaces. 2016; 148: 474-480. [CrossRef]
- Hanson MS, Park EE, Sears ML, Greenwood KK, Danobeitia JS, Hullett DA, et al. A simplified approach to human islet quality assessment. Transplantation. 2010; 89: 1178-1188. [CrossRef]
- Loganathan G, Graham ML, Radosevich DM, Soltani SM, Tiwari M, Anazawa T, et al. Factors affecting transplant outcomes in diabetic nude mice receiving human, porcine, and nonhuman primate islets: analysis of 335 transplantations. Transplantation. 2013; 95: 1439-1447. [CrossRef]
- Sweet IR, Gilbert M, Scott S, Todorov I, Jensen R, Nair I, et al. Glucose-stimulated increment in oxygen consumption rate as a standardized test of human islet quality. Am J Transplant. 2008; 8: 183-192.
- Papas KK, Colton CK, Nelson RA, Rozak PR, Avgoustiniatos ES, Scott WE, et al. Human islet oxygen consumption rate and DNA measurements predict diabetes reversal in nude mice. Am J Transplant. 2007; 7: 707-713. [CrossRef]
- Ichii H, Inverardi L, Pileggi A, Molano RD, Cabrera O, Caicedo A, et al. A novel method for the assessment of cellular composition and beta-cell viability in human islet preparations. Am J Transplant. 2005; 5: 1635-1645. [CrossRef]
- Armann B, Hanson MS, Hatch E, Steffen A, Fernandez LA. Quantification of basal and stimulated ROS levels as predictors of islet potency and function. Am J Transplant. 2007; 7: 38-47. [CrossRef]
- Suszynski TM, Wildey GM, Falde EJ, Cline GW, Maynard KW, Ko N, et al. The ATP/DNA ratio is a better indicator of islet cell viability than the ADP/ATP ratio. Transplant Proc. 2008; 40: 346-350. [CrossRef]
- Mohammed JS, Wang Y, Harvat TA, Oberholzer J, Eddington DT. Microfluidic device for multimodal characterization of pancreatic islets. Lab Chip. 2009; 9: 97-106. [CrossRef]
- Adewola AF, Lee D, Harvat T, Mohammed J, Eddington DT, Oberholzer J, et al. Microfluidic perifusion and imaging device for multi-parametric islet function assessment. Biomed Microdevices. 2010; 12: 409-417. [CrossRef]
- Nourmohammadzadeh M, Xing Y, Lee JW, Bochenek MA, Mendoza-Elias JE, McGarrigle JJ, et al. A microfluidic array for real-time live-cell imaging of human and rodent pancreatic islets. Lab Chip. 2016; 16: 1466-1472. [CrossRef]
- Nourmohammadzadeh M, Lo JF, Bochenek M, Mendoza-Elias JE, Wang Q, Li Z, et al. Microfluidic array with integrated oxygenation control for real-time live-cell imaging: effect of hypoxia on physiology of microencapsulated pancreatic islets. Anal Chem. 2013; 85: 11240-11249. [CrossRef]
- Xing Y, Nourmohammadzadeh M, Mendoza Elias JE, Chan M, Chen Z, McGarrigle JJ, et al. A pumpless microfluidic device driven by surface tension for pancreatic islet analysis. Biomed Microdevices. 2016; 18: 80. [CrossRef]
- Resto PJ, Berthier E, Beebe DJ, Williams JC. An inertia enhanced passive pumping mechanism for fluid flow in microfluidic devices. Lab Chip. 2012; 12: 2221-2228. [CrossRef]
- Berthier E, Beebe DJ. Flow rate analysis of a surface tension driven passive micropump. Lab Chip. 2007; 7: 1475-1478. [CrossRef]



